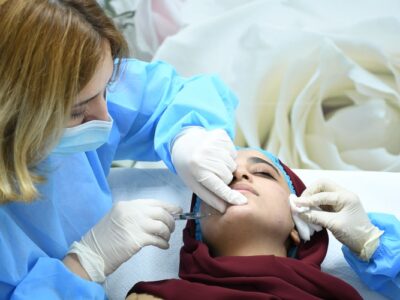
The global race to develop an effective vaccine against the novel coronavirus has resulted in the creation of numerous innovative solutions. One such breakthrough is the Zydus Needle-Free Corona Vaccine ZyCoV-D. Developed by Ahmedabad-based pharmaceutical giant Zydus Cadila, ZyCoV-D is the world’s first DNA-based vaccine against SARS-CoV-2 and has received Emergency Use Authorization (EUA) in India. In this comprehensive article, we will explore the unique features of ZyCoV-D, its efficacy, safety, and manufacturing process, as well as its potential impact on the global fight against COVID-19.
What is ZyCoV-D?
ZyCoV-D is a plasmid DNA vaccine that generates an immune response against the SARS-CoV-2 virus. The vaccine works by using a non-replicating variant of a DNA molecule known as plasmid to help synthesize a safe version of the spike protein found on the SARS-CoV-2 membrane. DNA-based vaccines, also known as third-generation vaccines, use modified DNA to trigger the immune system to fight against the virus. The World Health Organization (WHO) claims this “radical new approach” has several benefits over conventional vaccines, including improved vaccine stability and the absence of any infectious agent.
How ZyCoV-D Differs from Other COVID-19 Vaccines
While all COVID-19 vaccines aim to protect against the virus, ZyCoV-D differs significantly from other intramuscular vaccines currently available. Some of the key differences include:
Three-Dose Regimen
In contrast to other COVID-19 vaccines in India, ZyCoV-D requires three doses to be administered on days 0, 28, and 56. Zydus Cadila is also evaluating a potential two-dose regimen for the vaccine.
Needle-Free Administration
ZyCoV-D is a needle-free vaccine that uses a “Jet Injector” to administer the doses. The vaccine is delivered directly through the top layer of skin and into the tissues underneath using high pressure, instead of injecting the medicine by puncturing the skin with a needle. This needle-free method can significantly reduce the risk of side effects associated with intramuscular injections, such as arm soreness, redness, and pain at the injection site.
DNA-Based Vaccine
ZyCoV-D is the first plasmid DNA vaccine approved for use in humans. DNA vaccines have the potential advantage of being easily modified to protect against viruses that are constantly evolving.
Efficacy of ZyCoV-D
According to the interim assessment from Phase 3 clinical trials involving more than 28,000 volunteers, ZyCoV-D has demonstrated an efficacy rate of 66.6% for symptomatic RT-PCR positive cases. After the third dose of the vaccine, it was observed that none of the vaccine recipients had mild cases of COVID. Furthermore, no severe diseases or deaths were reported among those who received subsequent doses. However, the phase three trial’s data is still pending peer review.
Safety and Age Group Suitability
ZyCoV-D is the first vaccine candidate found to be suitable for children aged 12 and above. The vaccine has also been tested on teenagers between the ages of 12 and 18, marking the first time any COVID-19 vaccine has been trialed on this age group in India. Another vaccine candidate, Covaxin from Bharat Biotech, is currently undergoing trials for those aged 2 to 18.
Manufacturing and Availability
Zydus Cadila has announced that it will produce 10-12 crore doses of ZyCoV-D per year. The company is also exploring potential partnerships to increase production by an additional 60-70 million doses, with a target of 200 million doses annually. The vaccine is expected to be available within 45 to 60 days of authorization, pending necessary approvals and production increases.
Current Manufacturing Capacity
Zydus Cadila’s new ZyCoV-D manufacturing plant is scheduled for completion by the end of July. Additionally, at its current facilities, it can manufacture 4.5 million doses.
Potential Partnerships
To further increase production capacity, Zydus Cadila is exploring potential partnerships with other pharmaceutical companies and contract manufacturing organizations, such as Shilpa Medicare Ltd and Enzychem Lifesciences of the Republic of Korea.
Pricing and Distribution
The pricing of ZyCoV-D has yet to be announced. Zydus Group Managing Director Sharvil Patel has stated that the company will work closely with regulatory authorities to determine the pricing and modality of delivery of the vaccine doses. Clarity on pricing is expected in the coming weeks.
Emergency Use Authorization and Regulatory Approvals
Zydus Cadila submitted an Emergency Use Authorization (EUA) application for their ZyCoV-D three-dose COVID-19 vaccine on July 1, 2021. The Drugs Controller General of India (DCGI) granted EUA to ZyCoV-D on August 20, 2021, making it the sixth vaccine to be approved for use in India.
Approval for Two-Dose Regimen
Zydus Cadila has also received permission from the drug regulatory authority to conduct Phase III trials for a two-dose regimen of ZyCoV-D. The company believes that the two-dose regimen could help vaccinate more people in a shorter time and improve overall immunity against the virus.
Impact on India’s Vaccination Drive
ZyCoV-D is expected to play a significant role in India’s vaccination drive, especially in vaccinating the younger population. With the approval of ZyCoV-D for children aged 12 and above, India now has three vaccines approved for use in this age group, along with Zydus Cadila’s DNA shot ZyCoV-D and homegrown player Bharat Biotech’s Covaxin.
Inclusion in Government Vaccination Program
Zydus Cadila’s COVID-19 vaccine ZyCoV-D is expected to be included in the government’s national anti-coronavirus vaccination program, initially for adults and eventually for children aged 12 and above as well.
Conclusion
The Zydus Needle-Free Corona Vaccine ZyCoV-D represents a significant milestone in the global fight against COVID-19. Its unique features, such as the DNA-based technology, needle-free administration, and suitability for children aged 12 and above, make it a promising candidate to help control the pandemic. As production and distribution ramp up, ZyCoV-D has the potential to make a substantial impact on vaccination efforts in India and around the world.